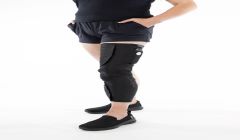
Cionic’s Neural Sleeve granted USFDA clearance
Neural sleeve first algorithm-powered bionic garment designed to help those with mobility issues due to multiple sclerosis, stroke, cerebral palsy, and other conditions, regain functional movement in everyday activities