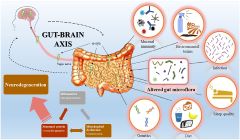
Alzamend Neuro appoints Dr. Terri Hunter to its Scientific Advisory Board
During her tenure at the University of South Florida Dr. Hunter was responsible for managing the patent portfolio associated with Alzamend’s two product candidates, AL001 and AL002