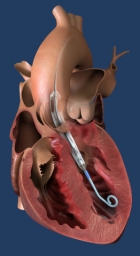
FDA grants breakthrough device designation to Impella ECP, the world’s smallest heart pump
It measures 9 French (3 millimetres) in diameter upon insertion and removal from the body. While in the heart, it expands to support the heart’s pumping function, providing flow greater than 3.5 L/min








_256_384.jpg)




