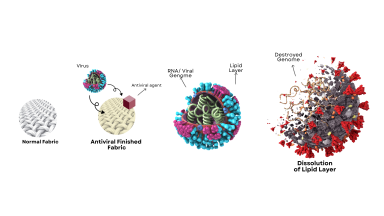
Glenmark Pharma and SaNOtize announce Peer Reviewed Publication of its Phase 3 Clinical Trials on SaNOtize's Novel Nitric Oxide Nasal Spray
The study demonstrated that patients who received NONS had significant reduction in viral load within 24 hours, which was sustained over seven days of treatment.